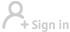Philippines approves China's Sinovac COVID-19 vaccine as booster shots

MANILA - The Philippines' Food and Drug Administration (FDA) has approved the use of China's Sinovac CoronaVac vaccine and three other brands as booster shots against the COVID-19, FDA Director General Enrique Domingo said Tuesday.
During a pre-recorded meeting aired on Tuesday, Domingo said the FDA has already amended the emergency use authorization of Sinovac, Pfizer, AstraZeneca and Sputnik Light, "meaning six months after the second dose, another can be administered."
The Philippines' Department of Health (DOH) said that all fully vaccinated healthcare workers could get the booster doses starting on Wednesday, as well as people with underlying health conditions and the elderly with comorbidities.
To date, the Philippines has received almost 125 million doses of COVID-19 vaccines from different vaccine makers. China, the biggest supplier of COVID-19 vaccines, was the first country to provide vaccines to the Philippines, allowing the Southeast Asian country to kick off its vaccination drive on March 1.
The Philippines has administered nearly 71 million doses of COVID-19 vaccines. More than 31.8 million people have been fully vaccinated. The government aims to vaccinate up to 70 percent of its 110 million population this year.
As of Tuesday, the country has reported a total of nearly 2.82 million confirmed cases of COVID-19, including 45,808 deaths.