China's new recombinant COVID-19 vaccine starts human tests

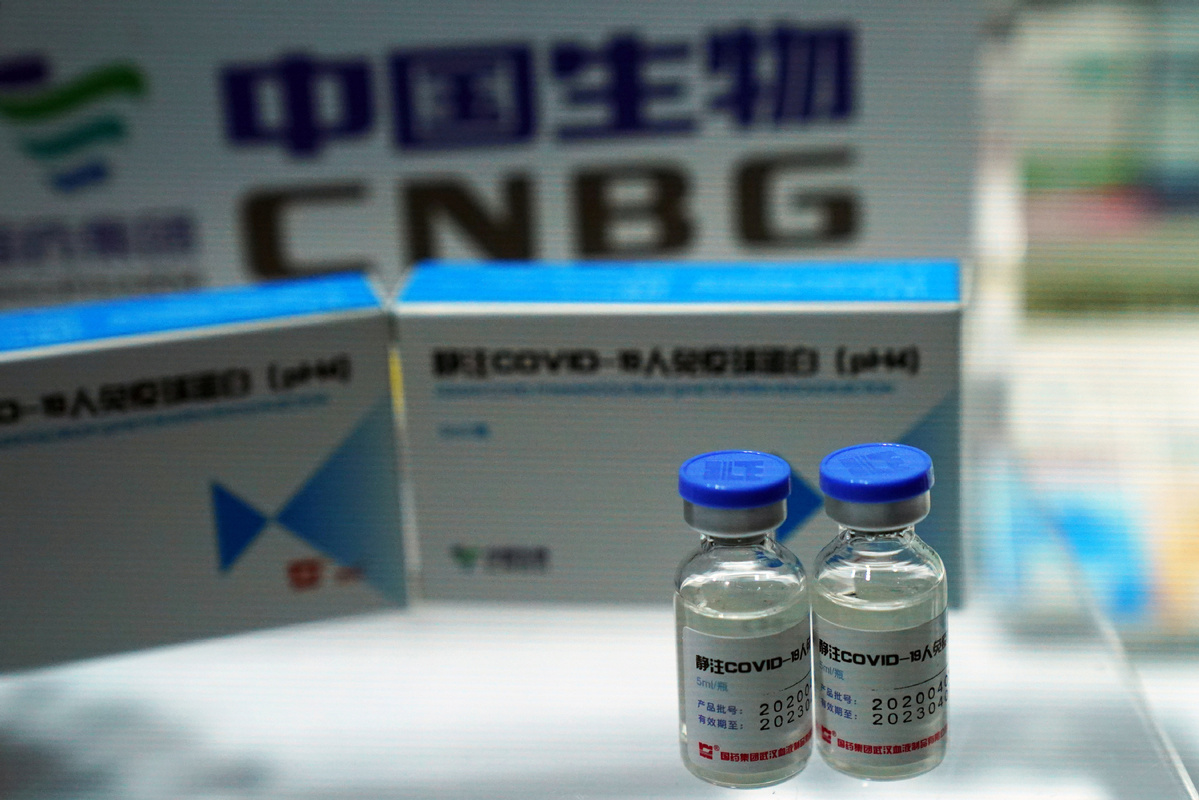
BEIJING -- Chinese pharmaceutical giant Sinopharm has commenced phase-1 and phase-2 clinical trials for its new recombinant vaccine against COVID-19 in Central China's Henan province since Saturday.
Developed by the China National Biotec Group (CNBG), a Sinopharm's bioscience subsidiary, the vaccine obtained a clinical research permit from the National Medical Products Administration on April 9.
This is CNBG's third COVID-19 vaccine candidate approved for human trials.
Researchers started the early-staged clinical trials in the city of Shangqiu, Henan Province. The trials were randomized, double-blind and placebo-controlled, according to a company statement.
Zhang Yuntao, vice president of the CNBG, said the recombinant vaccine candidate has been genetically engineered. Unlike the company's inactivated COVID-19 vaccines, the new one does not need a high-grade biosafety laboratory for manufacture, and it can quickly achieve large-scale production.
"It is designed for people aged three and above," Zhang said.
Whether the gene recombinant vaccine should be administered in three doses will be determined by the results of clinical trials, Zhang added.
- Ultra-cheap dress blind boxes spark health, quality concerns
- Chinese researchers find new treatment path for high-risk breast cancer
- China cracks down on organized crime involving minors
- Two Taiwan suspects wanted in mainland smuggling case
- Lhasa promotes initiative to foster a skilled workforce
- Beijing makes it easier for families to buy property